Are Microbiome Therapeutics Finally Here?
What do the recent FDA approvals tell us about the state of the field?
Quick Summary
Microbiome as a therapeutic modality has notched some big wins over the past six months, with two microbiome therapeutic FDA approvals (Rebyota by Ferring & Vowst by Seres).
The approvals thus far are a boon for patients, but do not provide a conclusive proof-point for many of the most risky challenges facing the microbiome field.
The field is still looking for later-stage clinical evidence and approvals that demonstrate our ability to identify targets in the gut, to select or engineer strains for treatment, and to expand to indications outside of infectious disease.
In just six months, the FDA approved the first two microbiome-based therapeutics ever in the United States with Ferring’s Rebyota and Seres Therapeutics’s Vowst. These are major milestones for a field which has been hungry for clinical and commercial proof-points.
Prior to these approvals, the field has weathered notable setbacks in recent years both in the clinic (see Seres’s own SER-109 and SER-287 pivots) as well as in the boardroom (see the shut-downs of early leaders like Kaleido & Finch Therapeutics). Large pharma has also pulled back from the space with leaders such as Takeda deprioritizing pharma programs.
Given that recent backdrop, these approvals are welcome signs of progress and traction in the space. However, it’s important to clarify what exactly these approvals tell us about the state of the field and our grasp of the microbiome as a tool for preventing and curing disease.
In the field of the microbiome, we are still looking for clinical proof-points demonstrating that we can actually understand our gut biology (and the biology of the trillions of microbes within our gut) to identify the right disease targets, that we can discover or engineer microbial medicines to hit those targets, and that we have the ability to grow microbes that typically live in your colon in commercial-scale infrastructure. Further, the field of microbiome still needs to demonstrate that the microbiome can be an effective lever not just for infectuous disease, but modularly across indications ranging from autoimmune, to mental health, to CNS diseases, to rare disease, and more. What do the recent approvals say, if anything, about our ability to rise to these challenges?
A Brief History of Microbiome Medicine
Stage 0 - The Fecal Microbiome Transplant
In the 1970s, doctors first began exploring fecal microbiome transplants (FMT) as a tool to fight recurrent C. diff infections (although records exist of an oral FMT procedure used in 4th century China). C. diff are bacterial infections that primarily affect individuals with weakened immune systems (often seen in patients on antibiotics after prolonged hospital stays) causing severe diarrhea and potentially life-threatening inflammation of the colon. When the good bacteria in our gut are depleted (say, by broad spectrum antibiotics), it opens up space in the gut for bad bacteria, like C. diff, to move in.
For the past few decades, fecal transplants have taken poop from screened, healthy donors and transplanted that poop into C. diff patients. This decades-old brand of poop transplant - despite the ‘ick’ factor - is highly efficacious, resolving symptoms in 85% or more of patients.
FMT has been regulatorily a bit of an anomaly, as the FDA has practiced ‘enforcement discretion’ since 2013 and essentially decided not to regulate FMT as a drug. Today, FMT is practiced widely, but is largely left unregulated.
Stage 1 - The First Microbiome Therapeutic Approvals
Over the past decade or so, a wave of microbiome therapeutic companies have tried to push microbiome medicine into the future with standardized, efficacious, safe, and targeted therapeutics that expand far beyond infectious disease and C. diff infections. The field has finally achieved its first couple of regulatory wins, with FDA approvals for two products granted since November 2022.
In reality, the modality of the microbiome is actually composed of many different ‘sub-modalities’ ranging from untargeted consortia of microbes, to ‘designed’ consortias, to single-strain microbes, to genetically-engineered bugs, to small-molecule microbiome modulators. Across each of these branches of microbial medicines, the field grapples with a common set of risks ranging from our ability to actually understand the complex biology of our gut, to our ability to select or engineer effective microbial drugs, and to our ability to actually manufacture these funky bugs which are more used to growing in our guts than in a GMP facility.
So, before we conclude that the Era of the Microbiome is upon us, let’s look into what these approvals have actually de-risked in the field.
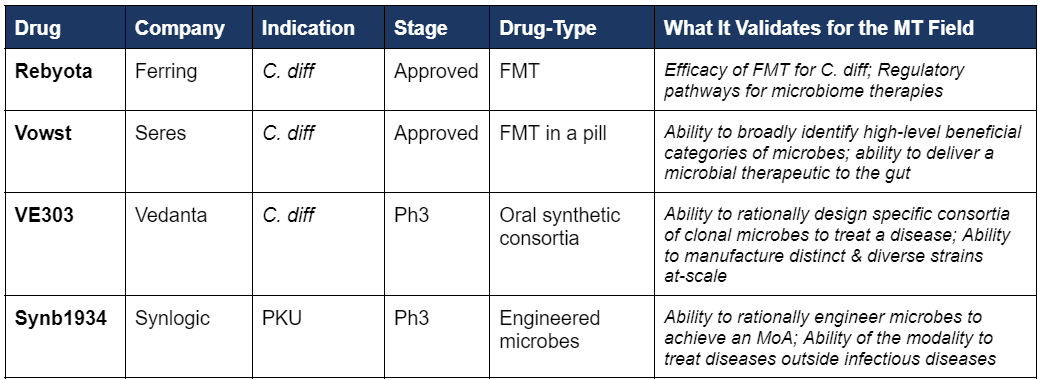
Rebyota by Ferring: In late 2022, the FDA approved the first microbiome-based therapeutic, Rebyota by Ferring, to treat C. diff. Rebyota is essentially a ‘screened & cleaned’ supply of poop slurry for FMTs. Rebyota is produced from donor samples, which are then screened and processed into a liquid mixture which can be administered to patients via enema in a similar manner to FMT. While the approval is a boon for patients and should make accessing FMTs easier and safer, the Rebyota approval did not provide a real proof-point to counter the hardest critiques against the microbiome field. For example, Rebyota did not demonstrate the field’s ability to understand the Mechanism of Action behind the microbiome, to select or engineer efficacious microbes or consortia, or even our ability to grow and manufacture microbial therapeutics. The Rebyota approval is a win for patients and for demonstrating the regulatory pathway for microbiome therapeutics, but did not de-risk the microbiome target identification, product engineering, or microbial manufacturing challenges faced by others in the space.
Vowst by Seres: Vowst, developed by Seres, received approval for C. diff this past week from the FDA and took the microbiome therapeutics field one step further into the future. It represents an extra step of validation past Rebyota by (1) shifting the administration from a transplant to a pill, and by (2) incorporating some high-level elements of ‘design’. If Rebyota is simply ‘screened & cleaned’ fecal samples, Vowst incorporates some amount of ‘processing’ to enrich the fecal samples to contain higher amounts of a broad microbial phylum, the spore-forming Firmicutes. However, enriching for a broad phylum of microbes is a far cry from selecting specific clonal strains, and there is still considerable variation in product composition between batches and donors. From a manufacturing perspective, Vowst is still dependent on fecal donor samples, which are then screened and treated to enrich the proportion of Firmicutes spores. Vowst is a first-step gesture to the field’s ability to understand the mechanism of action behind gut-based therapeutics and to select beneficial microbes; however, it does not demonstrate our ability to ‘engineer’ microbial consortia intelligently de novo and it does not de-risk the manufacturing challenges inherent in producing consortia of specific, diverse bugs.
Stage 2: Looking forward
Looking forward, we will hope to see clinical data and regulatory approvals that give proof-points for some of the as-yet-less-validated elements of microbiome-based therapies. These could include therapies that demonstrate an ability to:
Rationally Pick Strains & Consortia: In contrast to Rebyota and Vowst which are sourced by processing fecal samples, future therapies will have carefully designed consortia consisting of only specific microbes. Vedanta’s VE303, which just raised over $100M to progres its lead program, represents just such a drug, a rationally picked consortia of 8 microbes to treat C. diff. This will be an important moment to demonstrate that the field is far enough along to identify specific strains that can provide therapeutic impact.
Efficiently Manufacture Diverse Consortia: Human gut commensals are often not the easiest bugs to culture and manufacture at-scale, often requiring unique anaerobic conditions or nutrient feeds. Companies like Vedanta and others have navigated these challenges through Ph2-trial scale, but this will continue to be an area to monitor particularly if therapies are taking on large-population indications (i.e., IBD).
Indication Variety: Both microbiome approvals to date have been for infectious disease (i.e., C. diff), and we’re still waiting for the approvals of drug candidates that demonstrate we understand enough about the gut to develop medicines for indications outside of infectious disease, particularly in areas such as autoimmune disease (i.e., IBD) and the gut-brain-axis.
Engineered Strains: An engineered strain designed to perform a specific task in the gut has yet to gain approval, but promising therapies are now entering Ph3 trials, including Synlogic’s PKU candidate.
We’ve already seen the start of these proof-points as products have progressed through preclinical and early-stage clinicals, and the upcoming years hold many more opportunities for validation in later-stage trials. With these proof-points in-hand, microbiome therapeutics could be poised to emerge as a multi-tool modality with a broad impact across diverse indications ranging from infectious disease, to autoimmune, to mental health and more.
Hello! My name’s Matt Kirshner, and I’m an investor and incubation lead at Ferment, a venture studio building product-focused companies on top of the world’s most powerful biotechnology platforms. Previously, I worked across multiple roles at Ginkgo Bioworks and as a management consultant in the biotechnology space at Putnam Associates.
As a disclaimer, the views expressed here do not reflect those of anyone but my own.